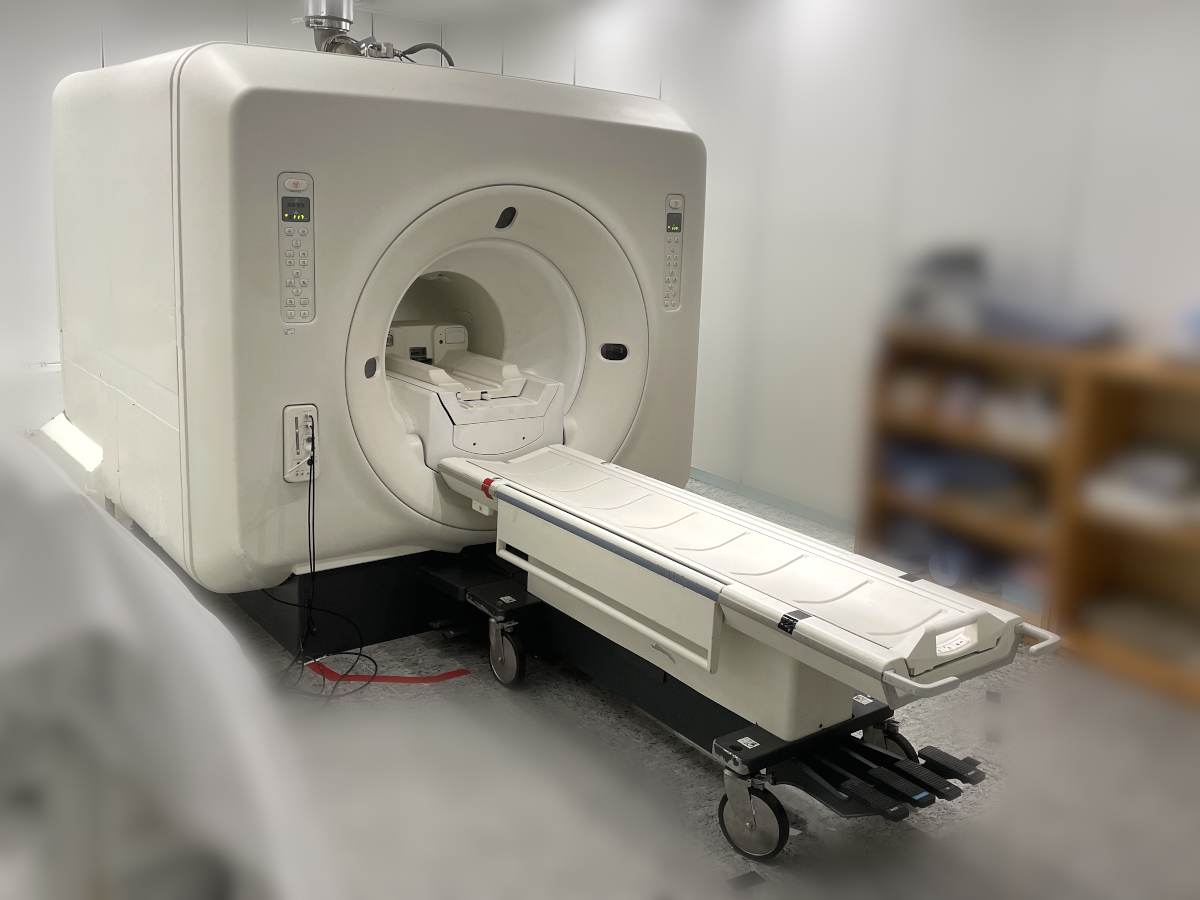
Primary use
Type of medical device or related product
Multiple vs. specific use (health condition)
Intended population age
Intended population sex
Level of technical knowledge
Capital
Reusable
Requirements
air conditioning / temperature control
electricity (mains)
emergency power supply
Fire detection and protection
HIS/LMIS/RIS/PACS
lighting
reinforced floor or ceiling
water (cold)
WHO list of priority medical devices
Service delivery platforms / Healthcare levels
Healthcare unit
EMDN related code(s)
Z110501
MAGNETIC RESONANCE (MR) SYSTEMS
The code(s) and term(s) in this section were observed and retrieved from public databases and have not been validated by health regulatory authorities.
Please consult your regulatory agency and EMDN site:
https://webgate.ec.europa.eu/dyna2/emdn
GMDN related code(s)
37654
Full-body MRI system, superconducting magnet (A diagnostic general-purpose magnetic resonance imaging (MRI) system designed to scan any targeted area of the body (full-body imaging). This system includes a superconducting magnet assembly and can be fixed-location, mobile, or transportable. Some systems can perform MR spectroscopy or various real-time imaging procedures for MRI guided interventional, therapeutic, or surgical applications. The system is available in a variety of system gantry configurations including closed bore, open bore, and open-sided or other kinds of patient accessible designs.)
The medical device term(s), code(s) and definition(s) in this section were retrieved from databases external to WHO.
As there might be more than one name, definition and “Nomenclature Code” related to the specific medical device, please consult
https://gmdnagency.org
GMDN ®. © GMDN Agency 2005-2025
WHO related resources
WHO resources on medical devices
Interagency list of priority medical devices for essential interventions for reproductive, maternal, newborn and
child health
WHO list of priority medical devices for cancer management
WHO List of Priority Medical Devices for management of cardiovascular diseases and diabetes
WHO List of Priority medical devices list for the COVID-19 response and associated technical specifications
Package of eye care interventions
Trauma and Emergency Surgery Kit (TESK) 2019
WHO medical devices website
WHO priority medical devices website
WHO essential in vitro diagnostic website


