Primary use
Type of medical device or related product
Multiple vs. specific use (health condition)
Intended population age
Intended population sex
Level of technical knowledge
Capital
Reusable
Requirements
electricity (mains)
WHO list of priority medical devices
Service delivery platforms / Healthcare levels
Healthcare unit
EMDN related code(s)
W0202050303
OPTICAL LABORATORY MICROSCOPES
The code(s) and term(s) in this section were observed and retrieved from public databases and have not been validated by health regulatory authorities.
Please consult your regulatory agency and EMDN site:
https://webgate.ec.europa.eu/dyna2/emdn
GMDN related code(s)
65539
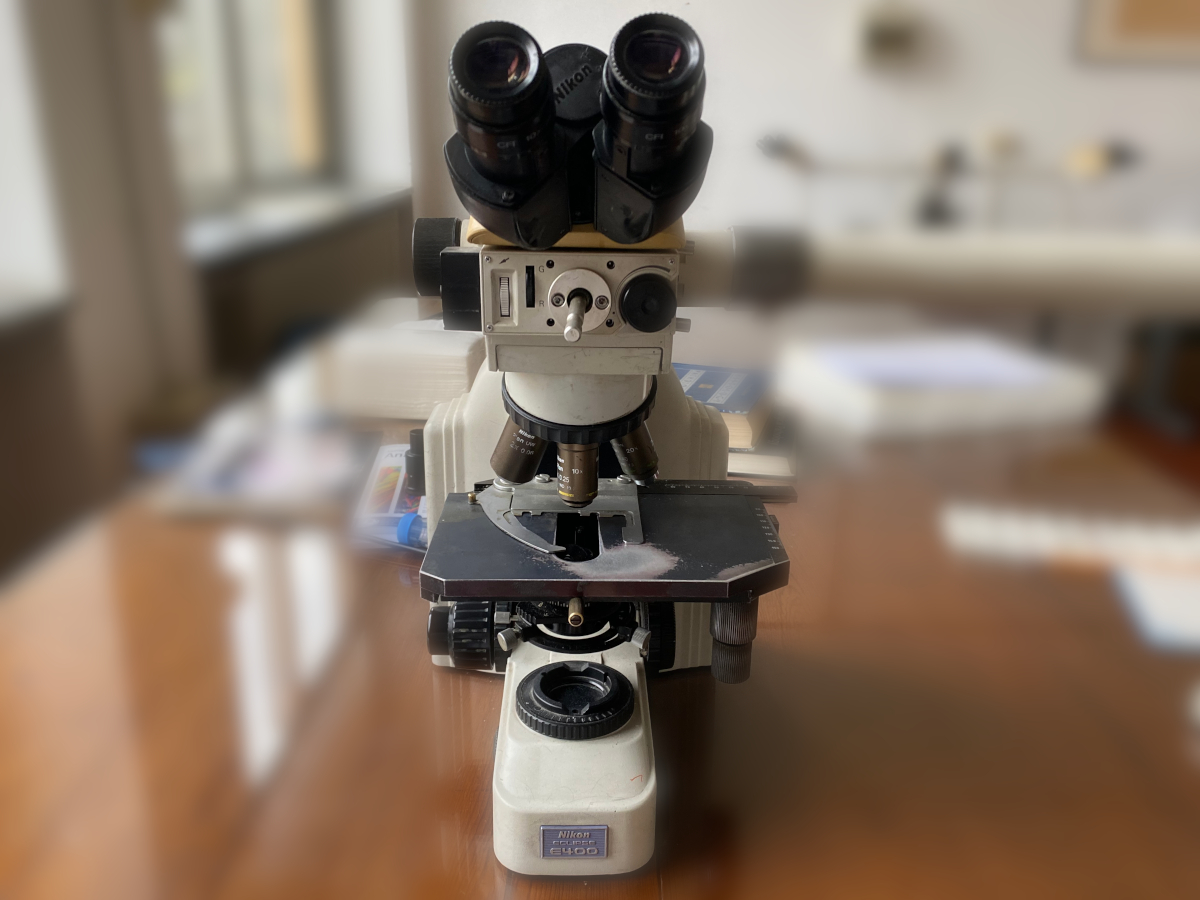
Basic light microscope IVD (A dedicated in vitro diagnostic magnification instrument designed with a standard microscope configuration [the object viewed through an ocular(s) from above] used to magnify and study specimens and small objects by transmitted visible light. It is widely used in laboratories and for research and in healthcare facilities where there is a need for such magnification. The device consists of a stand that supports the main components, an ocular body in which the eyepieces are mounted, a nosepiece to support the objectives, a specimen stage, a condenser to focus the light on the specimen, and a source of light [e.g., tungsten (W), halogen] and light port.)
The medical device term(s), code(s) and definition(s) in this section were retrieved from databases external to WHO.
As there might be more than one name, definition and “Nomenclature Code” related to the specific medical device, please consult
https://gmdnagency.org
GMDN ®. © GMDN Agency 2005-2025
WHO Technical specifications
WHO related resources
WHO resources on medical devices
Interagency list of priority medical devices for essential interventions for reproductive, maternal, newborn and
child health
WHO list of priority medical devices for cancer management
WHO List of Priority Medical Devices for management of cardiovascular diseases and diabetes
WHO List of Priority medical devices list for the COVID-19 response and associated technical specifications
Package of eye care interventions
Trauma and Emergency Surgery Kit (TESK) 2019
WHO medical devices website
WHO priority medical devices website
WHO essential in vitro diagnostic website
Related IVDs Technical Specifications available
Related Innovative Technologies
Related WHO Essential in vitro diagnostic test
Body fluids analysis
Cerebrospinal fluid analysis
Cytopathological Examination
ER/PR IHC test
Giemsa stain
Gram stain
Histopathological Examination
Human epidermal growth factor receptor 2 immunohistochemistry test
Immunohistochemistry (IHC)
Indian Ink
Kato-Katz faecal smear
Kleihauer-Betke acid-elution test
Lactophenol cotton blue
Modified Acid-Fast stain
Panel of immunohistochemical (IHC) markers for diagnosis of of hematologic malignancies
Panel of immunohistochemical (IHC) markers for diagnosis of solid tumours
Papanicolaou (Pap) stain
Peripheral blood film examination
Plasmodium spp.
Potassium hydroxide (KOH) stain
Reticulocyte count
Sodium metabisulfite slide test
Urine microscopic exam
Venereal disease research laboratory (VDRL) test
White blood cell differential count
Ziehl-Neelsen stain


